 There is a fraction of lysosomal LAMP-2A that resides in the lumen and is not accessible for substrate binding or translocation. Mb) microdomains, where it undergoes partial cleavage by cathepsin A, followed by rapid degradation of this truncated product in the lysosomal lumen (L. Under conditions of low CMA activity ( left), lysosome-associated membrane protein (LAMP)-2A is recruited to lysosomal membrane (L. Local regulation of chaperone-mediated autophagy (CMA) activity in lysosomes. Better molecular characterization of the different components of this pathway in recent years, along with the development of transgenic models with modified CMA activity and the identification of CMA dysfunction in different severe human pathologies and in aging, are all behind the recent regained interest in this catabolic pathway. Substrate proteins undergo unfolding and translocation across the lysosomal membrane before reaching the lumen, where they are rapidly degraded. 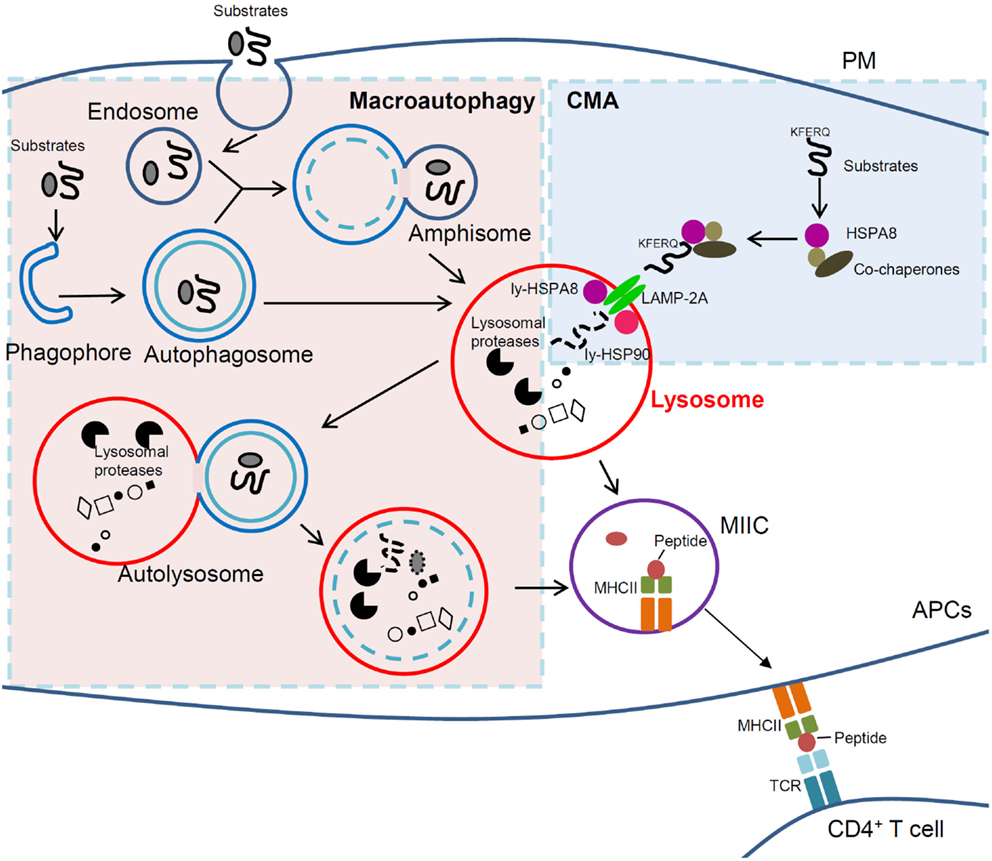
Selectivity in CMA is conferred by the presence of a targeting motif in the cytosolic substrates that, upon recognition by a cytosolic chaperone, determines delivery to the lysosomal surface. In this review, we focus on chaperone-mediated autophagy (CMA), a selective form of autophagy that modulates the turnover of a specific pool of soluble cytosolic proteins. Different pathways contribute to the degradation of intracellular components in lysosomes or autophagy. In fact, failure to timely turnover proteins and organelles leads often to cell death and disease. 
Continuous renewal of intracellular components is required to preserve cellular functionality. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
